
ここからコンテンツです。

Why can our brains learn and memorize?
Comprehensive understanding of expression mechanism of long-term potentiation and long-term depression of hippocampal excitatory synaptic strength - By Kouji Harada
The long-term potentiation (LTP) and long-term depression (LTD) of the strength of hippocampal excitatory synapse involved in learning and memory formation in the brain have been separately explained, but the molecular mechanism that comprehensively explains them has not been elucidated. Assistant Professor Dr. Kouji Harada, from the Department of Computer Science and Engineering at Toyohashi University of Technology and Associate Professor Dr. Tomonari Sumi, from the Research Institute for Interdisciplinary Science at Okayama University focused on the competition of exocytosis and endocytosis of AMPA-type glutamate receptors dependent on the number of calcium ions that flow in the postsynapse of hippocampal excitatory neurons, and demonstrated a comprehensive understanding of the LTP and LTD by a large-scale mathematical model simulation.
N-methyl-D-aspartate (NMDA)-type glutamate receptor (NMDAR)-dependent long-term potentiation (LTP) and long-term depression (LTD) in synapses on hippocampal excitatory neurons are considered to be a molecular basis essential to form the neural circuits involved in learning and memory. In mammalians, it has been proved that the main factor of the induction of LTP and LTD results in an increase and decrease in α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)-type receptor (AMPAR) at the postsynaptic membrane, depending on the calcium ion volume. However, the mechanism of the fluctuation of the AMPAR has not been elucidated. In addition, the true nature of the main pathway for AMPAR trafficking to the postsynaptic membrane remains up for debate as can be seen in the following studies.
Penn, et al. showed that the long-range lateral diffusion of AMPAR directed from areas other than the postsynaptic membrane (e.g. dendrite shaft) to the postsynaptic membrane is the main pathway for AMPAR trafficking for the LTP [1], and the long-range lateral diffusion pathway has been considered to be the most likely candidate as the main pathway. On the other hand, Wang et al. demonstrated the importance of the active transport of recycling endosomes containing AMPAR by molecular motor myosin Vb [2], and Wu et al. observed the exocytosis of the recycling endosomes containing AMPAR during the induction of LTP [3]. These studies embodied the elemental processes of AMPAR trafficking via the recycling endosome pathway. Currently, it is still unknown which pathway for AMPAR trafficking is the main one, but since all of these studies basically focus on the induction of LTP, there remains a need for an explanation of the pathway including the induction of LTD without inconsistency.
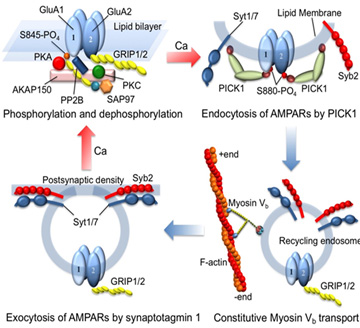
Assistant Professor Harada and Associated Professor Sumi modeled the following 4 processes of “active” transport of recycling endosomes containing AMPAR by molecular motor myosin Vb as a pathway for AMPAR trafficking to the postsynaptic membrane in order to comprehensively explain the LTP/LTD (Fig.1).
- AKAP150 signaling complex controlling the phosphorylation/dephosphorylation of subunits, GluA1 and GluA2, that constitute AMPAR (Upper left of Fig.1)
- Endocytosis of AMPAR to the cytoplasm by a calcium-binding protein, PICK1 (Upper right of Fig.1)
- Stationary active transport of recycling endosomes containing AMPAR toward postsynaptic membranes by myosin Vb (Lower right of Fig.1)
- AMPAR uptake at around postsynaptic membranes by Syt1/7-dependent exocytosis (Lower left of Fig.1)
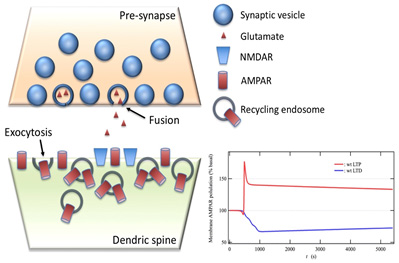
A simulation using a postsynaptic model based on these processes successfully reproduced the time course of the number of AMPAR corresponding to the induction of LTD and LTD observed in the experiment (Lower right of Fig. 2). In addition, the qualitative reproducibility of certain results demonstrated the validity of the model. These results include impaired LTP induction due to interference of Myosin Vb transport, impaired LTD induction due to decreased rate of reaction of PP2B-dependent dephosphorylation of AMPAR, impaired LTP and LTD inductions due to PICK1 expression level, and impaired LTP induction in Syt1 calcium-binding domain mutant.
The conclusions drawn from this simulation are as follows.
- The LTP (or LTD) expression is caused by a phenomenon whereby the exocytosis triggered by the activation of Syt1/7 becomes more predominant (or inferior) than the endocytosis triggered by PICK1 activation, resulting in an increase (or decrease) in the number of AMPAR at the postsynaptic membrane.
- The difference in calcium-dependent activation between the calcium sensors, PICK1 and Syt1, results in a difference in these calcium-binding constants.
- Myosin Vb carries the recycling endosomes containing AMPAR toward around the postsynaptic membrane by stationary ATP driven transport not dependent on calcium concentration.
- As a result, the recycling endosomes are waiting on cell membranes so that they can immediately address the next Syt1-dependent exocytosis, enabling the rapid LTP induction.
- AMPARs taken up to around the postsynaptic membrane due to exocytosis are immediately reallocated to the synapse membrane by the lateral diffusion.
The neural network model mimicking the higher brain function can learn changes of synapse coupling coefficient, and Hebbian rule is known as a most basic learning rule. The Hebbian rule and its extended/modified versions are currently used as learning rules, which are known to be closely related to NMDAR-dependent LTP. The achievement of this study provides a molecular basis for the Hebbian rule or changes of synapse binding, which is expected to be a hint to understand the high brain function from the molecular level.
This study was supported by the Grants-in-Aid for Scientific Research of Japan Society for the Promotion of Science (JSPS) (JP16K05657, JP18KK0151, JP16K00389).
References
[1]Penn AC, Zhang CL, Georges F, Royer L, Breillat C, Hosy E, et al. Hippocampal LTP and contextual learning require surface diffusion of AMPA receptors. Nature. Nature Publishing Group; 2017;549: 384–388. doi:10.1038/nature23658.
[2]Wang Z, Edwards JG, Riley N, Provance DW, Karcher R, Li X-D, et al. Myosin Vb Mobilizes Recycling Endosomes and AMPA Receptors for Postsynaptic Plasticity. Cell. 2008;135: 535–548.doi:10.1016/j.cell.2008.09.057
[3]Wu D, Bacaj T, Morishita W, Goswami D, Arendt KL, Xu W, et al. Postsynaptic synaptotagmins mediate AMPA receptor exocytosis during LTP. Nature. Nature Publishing Group; 2017;544: 316–321.doi:10.1038/nature21720.
Tomonari Sumi, Kouji Harada (2020). Mechanism underlying hippocampal long-term potentiation and depression based on competition between endocytosis and exocytosis of AMPA receptors. Scientific Reports, 10:14711, DOI: 10.1038/s41598-020-71528-3
なぜ私たちの脳は学習や記憶をすることができるのか
海馬興奮性シナプスにおける長期増強/長期抑制発現機構の統一的理解By 原田 耕治
これまで、脳の学習や記憶の形成に関わる海馬興奮性シナプスの長期増強(LTP)と長期抑制(LTD)はそれぞれ個別に説明が試みられてきましたが、それらを統一的に説明する分子機構は未解明でした。豊橋技術科学大学情報・知能工学系 原田耕治助教と岡山大学異分野基礎科学研究所 墨智成准教授は、海馬興奮性ニューロンの後シナプスに流入するカルシウムイオンの多寡に依存したAMPA型グルタミン酸受容体のエキソサイトーシスとエンドサイトーシスの競合に注目することで、LTPおよびLTDを統一的に理解できることを大規模数理モデルシミュレーションにより実証しました。
海馬興奮性ニューロンのシナプスにおけるNMDA型グルタミン酸受容体(NMDAR)依存長期増強(LTP)および長期抑制(LTD)は、学習や記憶に関わる神経回路形成に不可欠な分子基盤であると考えられています。哺乳類においては、LTPおよびLTD誘導の主要な要因は、カルシウムイオン量に応じたシナプス後膜でのAMPA型グルタミン酸受容体(AMPAR)の増加および減少に帰着することが確かめられています。しかしながら、その増減の機序は未だに解明されていません。さらに、シナプス後膜へのAMPAR輸送経路に関しても、主要な経路を巡る以下の論争があります。
Pennらは、シナプス後膜以外の領域(例えば樹状突起シャフト)からシナプス後膜へ向かうAMPARの長距離側方拡散が、LTPにおける主要なAMPAR供給経路である事を示し[1]、近年では長距離側方拡散経路が最も有力な候補として考えられる様になりました。ところが一方、WangらはミオシンVb分子モーターによるAMPAR含有再循環エンドソームの能動的輸送の重要性を示し[2]、またWuらは、LTP発現中にAMPAR含有再循環エンドソームのエキソサイトーシスが起きることを観測しました[3]。これらの研究により、再循環エンドソーム経路におけるAMPAR供給素過程がより具体化されました。現在、どちらのAMPAR輸送経路が主経路であるのかまだ確定していませんが、この主経路論争は基本的にLTP誘導に関するものであるため、LTD誘導を含めて矛盾無く説明できることが望ましいと考えられてきました。
原田助教と墨准教授はLTP/LTDを統一的に説明するため、シナプス後膜へのAMPAR供給経路として、ミオシンVb分子モーターによるAMPAR含有再循環エンドソームの「能動的」輸送を以下の4つのプロセスとしてモデル化しました(図1)。
- AMPARを構成するサブユニットGluA1とGkuA2のリン酸化/脱リン酸化を制御するAKAP150シグナル伝達複合体(図1左上)
- カルシウム結合タンパク質PICK1によるAMPARの細胞質へのエンドサイトーシス(図1右上)
- AMPAR含有再循環エンドソームのミオシンVbによるシナプス後膜方向への定常的能動輸送(図1右下)
- Syt1/7によるエキソサイトーシスによるAMPARのシナプス後膜周辺への取り込み(図1左下)
これらに基づく後シナプスモデルを用いたシミュレーションを実行し、実験で観測されているLTPおよびLTD誘導に対応するAMPAR数の時間変化を再現することに成功しました(図2右下)。加えて、ミオシンVb輸送の阻害によるLTP誘導の減少、PP2BによるAMPARの脱リン酸化反応率の低下によるLTD誘導の減少、PICK1発現量の減少によるLTPおよびLTD誘導の減少、並びにSyt1カルシウム結合ドメイン変異体におけるLTP誘導の減少等、報告されている観測結果を定性的に再現できることを示し、モデルの妥当性を実証しました。本シミュレーションから導かれた結論は以下の通りです。
- LTP(LTD)の発現は、Syt1/7活性化によるエキソサイトーシスがPICK1活性化によるエンドサイトーシスより優位(劣位)になり、シナプス後膜上AMPAR数が増加(減少)することに起因する。
- カルシウムセンサPICK1およびSyt1のカルシウム依存活性化の差は、これらのカルシウム結合定数の違いに帰着する。
- ミオシンVbはカルシウム濃度に依存しない定常的なATP駆動輸送により、AMPAR含有再循環エンドソームをシナプス後膜周辺方向へ運搬している。
- その結果として、次回のSyt1依存エキソサイトーシスに即時対応できるように、再循環エンドソームは細胞膜上で待機しており、迅速なLTP誘導を可能にしている。
- エキソサイトーシスによってシナプス後膜周辺へ取り込まれたAMPARは、側方拡散によりシナプス膜へ即座に再配置する。
脳の高次機能を模倣したニューラルネットワークモデルでは、シナプス結合係数の変化が学習に対応しており、最も基本的な学習則としてヘブ則が知られています。ヘブ則あるいはその拡張/変形したものが、現在でも学習則として用いられており、NMDAR依存LTPと密接に関係することが知られています。本研究成果はヘブ則あるいはシナプス結合の変化に対する分子基盤を与えており、分子レベルから脳の高次機能を理解する手がかりとなることが期待されます。
本研究は、独立行政法人日本学術振興会(JSPS)科学研究費補助金(JP16K05657、JP18KK0151、 JP16K00389)の助成を受け実施しました。
Researcher Profile

| Name | Kouji Harada |
|---|---|
| Affiliation | Department of Computer Science and Engineering |
| Title | Assistant Professor |
| Fields of Research | Theoretical Biology, Complex Systems |
ここでコンテンツ終わりです。
