
ここからコンテンツです。

Towards building next-generation batteries using a pigment electrode
Electrode design of next-generation batteries using Prussian blue and its analogues By Tomohiro Tojo
Tomohiro Tojo and his colleagues have demonstrated the properties of calcium ion batteries (CIBs) created using pigment electrodes such as Prussian blue and its analogues. The CIBs showed excellent cyclability of discharge and charge in a calcium-based organic electrolyte. This is thought to derive from strong atomic bonds in Prussian blue structures, which possess movable pathways for large-sized ions in three dimensions. Such unique structures have excellent potential for application in a new generation of batteries
Calcium ion batteries (CIBs) have attracted much attention as the next-generation batteries likely to replace lithium ion batteries (LIBs). This is because the theoretical capacity of CIBs is twice that of LIBs. This doubled capacity can be explained by the difference between monovalent and divalent ions. In addition, CIBs possess advantages such as lower cost and higher safety because calcium is more abundant than lithium and because CIBs have a higher melting point than LIBs. However, there is one major obstacle to the application of CIBs. It is hard to find a suitable electrode material in which calcium ions can be inserted and extracted reversibly because of the relatively large ionic radius of calcium ions (112 pm) as compared to that of lithium ions (76 pm).
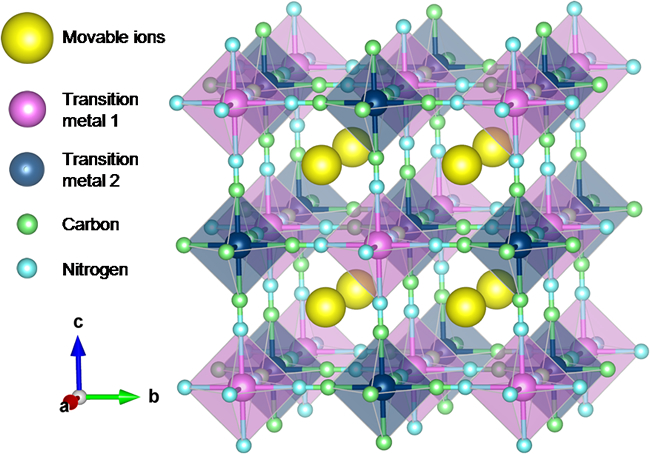
In this study, Tomohiro Tojo and his colleagues at the Department of Electrical and Electronic Information Engineering, Toyohashi University of Technology, employed Prussian blue (PB) and Prussian blue analogues (PBAs) as CIB electrodes because they possess large sites for inserting and extracting large-sized ions, as shown in Fig. 1. Up to now, using PBAs as an electrode material, the electrochemical behaviors of sodium ions corresponding to the radius of calcium ions have been examined in organic and inorganic electrolytes. These reports have shown reversible insertion and extraction of sodium ions into and from PBA structures.
The research team investigated the electrochemical performance of several PBA electrodes in order to determine whether calcium ions in an organic electrolyte exhibit either reversible or irreversible insertion and extraction into and from the crystal structure. Reversible capacities of 40–50 mAh/g were observed at a low current density, as shown in Fig. 2(a). In Fig. 2(b), the Coulombic efficiencies, which are defined as the ratio of the amount of insertion (discharge) and extraction (charge) of calcium ions, were observed to have a constant value of 90% after the 3rd cycle.
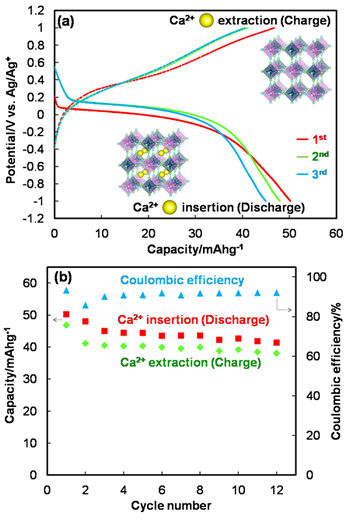
(a) Discharge-charge profiles of a Prussian blue analogue electrode in a calcium-based organic electrolyte and
(b) cycled capacities with discharge-charge capacity ratio
The results shown in Fig. 2 demonstrate the excellent cyclability of the PBA electrodes, even though the reversible capacities were half the theoretical capacity. The researchers investigated the reason for the high reversibility using X-ray diffraction (XRD) and X-ray photoelectron spectroscopy (XPS). The reversibility is explained by the durable structure of the PBAs and their excellent charge balance during the insertion and extraction of calcium ions.
This data suggests that Prussian blue (PB) and Prussian blue analogues (PBAs) may be proposed as feasible electrode materials for CIBs, although additional investigation is needed in order to enhance the reversible capacities still further. The researchers plan to carry out further study to improve the performance of CIBs relative to LIBs.
This work was partially supported by
- Grant-in-Aid for Challenging Exploratory Research (No. 15K13947) by MEXT.
- Grant-in-Aid for Scientific Research (B), No. 24360109 by MEXT
Reference
Tomohiro Tojo, Yosuke Sugiura, Ryoji Inada, and Yoji Sakurai (2016). Reversible Calcium Ion Batteries Using a Dehydrated Prussian Blue Analogue Cathode, Electrochimica Acta, 207, 22-27, 10.1016/j.electacta.2016.04.159
顔料電極を用いた次世代電池の構築に向けて
プルシアンブルー系電極を用いた次世代電池の設計電気・電子情報系 東城友都助教らの研究グループは、顔料であるプルシアンブルー(PB)・プルシアンブルー類似体(PBA)を電極材料に用いて、カルシウムイオン電池(CIB)の充放電試験を行いました。その結果、カルシウム系有機電解液中では、CIBの充放電サイクル性能が優れていることを示しました。これは、PBの原子同士が強固に結合しており、PBが大径のイオンを3次元的に伝導させるための経路を持つためであることが示唆されました。このような特異構造をもつ電極材料が次世代電池の開発に重要となります。
カルシウムイオン電池(CIB)は、理論上、リチウムイオン電池(LIB)の二倍の容量を示すため、LIBを超える次世代電池として注目を集めています。この容量の違いは、可動イオンに1価、2価のイオンを用いている違いによるものです。またCIBは、カルシウムの埋蔵量がリチウムに比べて多いことや融点が高いことから、LIBよりも低価格、高安全性な電池であることが考えられます。しかしながら、CIBの実現にはまだ問題点があります。それは、カルシウムイオン(112 pm)がリチウムイオン(76 pm)よりもイオン径が大きいために、カルシウムイオンを可逆的に挿入・脱離できる電極材料が少ないことが挙げられます。
そこで東城友都助教らの研究グループは、図1に示すように、大径のイオンを挿入・脱離することが可能とされるプルシアンブルー(PB)およびプルシアンブルー類似体(PBA)をCIB電極に採用しました。現在までにカルシウムイオンと同程度のイオン径であるナトリウムイオンを用いて、PBA電極の電気化学特性が有機電解液中・無機電解液中で評価されており、ナトリウムイオンの可逆的な挿入・脱離が確認されています。
また、ナトリウムイオンの代わりにカルシウムイオンを用いて、カルシウムイオンがPBAに可逆的に挿入・脱離するかどうかを調査するため、PBA電極の性能評価を行ないました。図2 (a)に示す通り、その充放電特性から40-50mAh/gの可逆容量を確認しました。また図2 (b)に示す通り、3サイクル以降のクーロン効率*は約90%で一定となりました。
図2の結果から、可逆容量は理論容量の半分程度ではあるが、クーロン効率すなわち、サイクル性能は優れていることが確認されました。このサイクル性能は何に起因しているのかX線回折(XRD)およびX線光電子分光法(XPS)により調査を行なった結果、PBAの壊れにくい構造と良好な電荷バランスに由来していることが判明しました。
本研究ではCIBに最適な電極材料として、プルシアンブルー(PB)およびプルシアンブルー類似体(PBA)を提案しましたが、PBA電極の可逆容量の向上については、更なる研究が必要です。今後は、LIBを超えるCIBの材料研究を引き続き進めていく予定です
*クーロン効率: クーロン効率は、充電容量(カルシウムイオン脱離量)/放電容量(カルシウムイオン挿入量)から求められる百分率で、この数値が100%に近い程、電池容量の損失が少ないことを示す。
本研究は文部科学省・科研費・挑戦的萌芽研究No. 15K13947の一部および、文部科学省・科研費・基盤研究(B) No. 24360109の支援を受けて遂行されました。
Researcher Profile

| Name | Tomohiro Tojo |
|---|---|
| Affiliation | Department of Electrical and Electronic Information Engineering |
| Title | Assistant Professor |
| Fields of Research | Energy Conversion Engineering / Electirical and Electronic Material Engineering |
ここでコンテンツ終わりです。
