
ここからコンテンツです。

Sodium solid electrolyte with high conductivity and electrochemical stability
Towards the development of all-solid-state sodium-ion batteries with long-term stability Hirotada Gamo
A research team led by Hirotada Gamo, a doctoral student in the Department of Electrical, Electronic and Computer Engineering at Toyohashi University of Technology, has developed Na3SbS4, in which part of the sulfur (S) is substituted with chlorine (Cl), as a solid electrolyte for all-solid-state sodium (Na) ion batteries. The ionic conductivity of Cl-substituted Na3SbS4 was improved by a factor of 3 at room temperature compared with that of the sample without Cl substitution. In addition, the crystal structure of Cl-substituted Na3SbS4 has a crystal structure framework that facilitates the three-dimensional migration of Na ions. Furthermore, the Cl-substituted Na3SbS4 showed better stability against Na anode.
The increasing demand for large-scale energy storage has stimulated research on developing all-solid-state sodium (Na) ion batteries using low-cost and abundantly available Na resources. The development of solid electrolytes that exhibit high ionic conductivity at room temperature is essential for the practical application of all-solid-state Na-ion secondary batteries. In order to meet this challenge, interest both in Japan and overseas has been focused on Na3SbS4 as the most promising candidate amongst the various solid electrolytes due to its high conductivity of 1 mS cm-1 or higher at room temperature. However, in order to achieve the high conductivity, post processing is required through ball milling, and achieving high ion conductivity through a simpler synthetic process has been notably problematic.
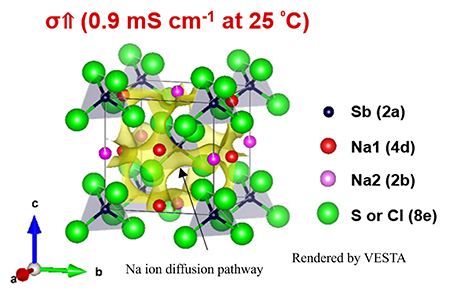
In response to this challenge, the research team lead by the Doctoral student Hirotada Gamo used a liquid-phase synthesis method suitable for mass production to develop a Cl-substituted Na3SbS4 solid electrolyte. By partially substituting S in the Na3SbS4 with Cl, the ionic conductivity at room temperature was enhanced by a factor of three (0.9 mS cm-1) compared to the sample without substitution (0.3 mS cm-1). In order to elucidate the effect of the structural change caused by the Cl substitution on the ionic conduction properties, the ionic conduction path was visualized by precise analysis of the crystal structure.
As a result, it was demonstrated that the partial substitution of Cl for S in Na3SbS4 results in the formation of a crystal structure framework in which Na ions are loosely bound to S (or Cl), i.e., there is a weak electrostatic interaction between Na and S (or Cl), which promotes ion diffusion, especially in the crystallographic c-axis direction. The increase in ionic conductivity by Cl substitution is attributed to the formation of a crystal structure with a three-dimensional ion diffusion pathway.
Additionally, the team discovered that the Cl-substituted Na3SbS4 solids exhibit better stability against the Na anode than the non-Cl substituted samples.. This improvement in electrochemical stability leads to a reduction in the interfacial resistance between the anode and the solid electrolyte, demonstrating that Cl substitution in large amounts is effective in improving the stability against the anode..
The research team has pointed the way towards future research by discovering important design guidelines for developing an ideal solid electrolyte with desirable characteristics such as high ionic conductivity and superior electrochemical stability. They believe that the solid electrolyte developed in this research could be combined with liquid-phase coating technology to realise all-solid-state Na-ion batteries with a high storage capacity and stable cycling.
Reference
Hirotada Gamo, Nguyen Huu Huy Phuc, Hiroyuki Muto, and Atsunori Matsuda, Effects of Substituting S with Cl on the Structural and Electrochemical Characteristics of Na3SbS4 Solid Electrolytes, ACS Applied Energy Materials 4(6), 6125-6134 (2021).
https://pubs.acs.org/doi/10.1021/acsaem.1c00927
高伝導性と電気化学安定性を兼ね備えたナトリウム固体電解質
長期安定な全固体ナトリウムイオン二次電池の開発に向けて
蒲生 浩忠
豊橋技術科学大学 電気・電子情報工学系の博士後期課程に在籍する蒲生浩忠をリーダとする研究チームは、全固体ナトリウム(Na)イオン二次電池用の固体電解質として、硫黄(S)の一部を塩素(Cl)置換したNa3SbS4を開発しました。Cl置換Na3SbS4は、Cl置換なしの試料と比較して、室温におけるイオン導電率が3倍まで向上しました。またCl置換したNa3SbS4の結晶構造は、Naイオンが3次元的に移動しやすい結晶構造フレームワークを持つことを実証しました。加えて、Cl置換はNa負極に対してより優れた安定性を示すことを見出しました。
大規模なエネルギー貯蔵に対する需要の高まりにより、低コストかつ埋蔵量の豊富なNa資源を用いた全固体Naイオン二次電池の研究が加速しています。全固体Naイオン二次電池の実用化には、室温で高いイオン伝導性を示す固体電解質の開発が必要不可欠です。Na固体電解質の中でも、Na3SbS4固体電解質は、室温で1 mS cm-1以上の高い導電率を示すことから、国内外で広く研究されています。しかしながら、その高い導電率の達成にはボールミリング処理による後処理が必要であり、より簡易な合成プロセスで高いイオン導電率を実現することが大きな課題となっていました。
そこで、蒲生浩忠らの研究グループは量産に適した液相合成法を用いて、Cl置換したNa3SbS4固体電解質を開発しました。Na3SbS4のSの一部をClで置換することで、置換なしの試料(0.3 mS cm-1)と比べて、室温でのイオン導電率が3倍(0.9 mS cm-1)に向上しました。また、Cl置換に伴う構造変化がイオン伝導特性に与える影響を解明するために、結晶構造の精密解析によりイオン伝導経路を可視化しました。
その結果、Na3SbS4中のSの一部をClで置換することで、NaイオンがS(またはCl)と局所的に緩く結合した、つまりNaとS(またはCl)との間で弱い静電相互作用を持つ結晶構造フレームワークが形成され、特に結晶学的なc軸方向へのイオン拡散が促進されることを実証しました。したがって、Cl置換に伴うイオン導電率の向上は、3次元的に開けたイオン拡散経路を持つ結晶構造が形成したことに起因します。
さらに、Cl置換したNa3SbS4固は、Cl置換なしの試料と比べて、Na負極に対して優れた安定性を示すことを見出しました。この電気化学的な安定性の改善は、負極と固体電解質の間の界面抵抗の低減に繋がり、Clの多量置換が負極に対する安定性の改善に効果的であることを実証しました。
研究チームは、高イオン伝導性や優れた電気化学的な安定性など望ましい特性を備えた理想的な固体電解質の開発に向けて、重要な設計指針を見出しました。また、本研究で開発した固体電解質と液相コーティング技術を組み合わせることにより、全固体Naイオン二次電池の高い蓄電容量および安定なサイクル特性を達成できると考えています。
Researcher Profile

| Name | Hirotada Gamo |
|---|---|
| Affiliation | Department of Electrical and Electronic Information Engineering |
| Title | Doctor course student |
| Fields of Research | Solid electrolyte/ sodium-ion battery/ Superionic conductor |
| Graduated KOSEN |
National Institute of Technology, Asahikawa College |
ここでコンテンツ終わりです。
