
ここからコンテンツです。

Sulfur improves birefringence!
Developing liquid crystalline molecules for high quality image displays By Yuki Arakawa
Yuki Arakawa, Assistant Professor of Toyohashi University of Technology, and his research team, has successfully liquid crystallized π-conjugated2) rod-like molecules with alkylthio groups containing sulfur1), and developed high birefringence molecules that exhibit nematic liquid crystal with high fluidity in temperature ranges including room temperature. This molecular design is expected to offer a new liquid crystal material that contributes to the high quality image resolution of liquid crystal displays.
Liquid crystal materials with high birefringence and dielectric constant have been contributing to lowering the driving-voltage and improving the response speed of liquid crystal (LC) displays. Recently, various approaches have been taken to apply high birefringence LC materials to broadband circularly polarized light-reflecting films for brightness enhancement film, or to cholestric LC lasers for continuous oscillation.
In terms of commercialization of products, LC materials need to be developed by either forming LC phases at room temperature or fixing the orientation state of LC. However, improving birefringence and dielectric constant requires both an anisotropic molecular structure and electron richness, making a rise in the phase transition temperature (especially melting point) inevitable due to large intermolecular forces. In short, it is difficult to form a liquid crystalline state under room temperature.
Assistant Professor Yuki Arakawa and his team took interest in alkylthio groups (SCmH2m+1) that contain “sulfur”, a component of hot springs and one of the few surplus resources Japan has. Although alkylthio groups have high polarizability and are expected to be an effective substitutional group for birefringence improvement, only a few successful cases of rod-like molecules with alkylthio groups forming liquid crystals have been reported due to their difficulty to crystalize.
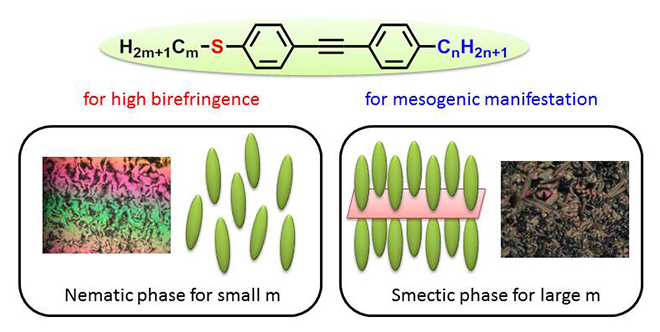
Yuki Arakawa and his team introduced substantially long alkyl chains (having with five or more carbons to one terminal of a diphenylacetylene structure 3) ) with to alkylthio groups to reveal that liquid crystallinity is exhibited during the cooling process. This is considered to be due to the fact that among the molecules aligned in an antiparallel configuration, long alkyl groups that prefer the out-of-plane conformation against the benzene ring inhibit molecular crystalline packing, and thus enable the molecules to rotate and translate while maintaining their orientation, which eventually leads to the formation of a liquid crystal phase.
Furthermore, the team observed a phenomenon where the melting point decreased due to the large bending and low electron donating properties of the alkylthio groups, and succeeded in developing a molecule that exhibits liquid crystallinity in temperature ranges including room temperature. Changing the carbon numbers in alkylthio groups after introducing long alkyl chains enables the formation of both a highly-ordered smectic phase with a high viscosity layer structure and a nematic (N) phase with low viscosity, which is particularly important for optical applications. Comparison with oxygen analogues4) confirmed significant improvement of optical properties. More recently, the team successfully developed new diphenyl-diacetylene-based π-conjugated liquid crystals with the maximum birefringence value of 0.3 or mesophases supercooled to room temperature, expanding the above mentioned molecular design.
"There were only a few reports on rod-like structure molecules with alkylthio groups exhibiting liquid crystalline phases, and no studies revealed the characteristics of these molecules, including the reason why they tend not to form liquid crystalline phases. We’re now aiming to utilize the characteristics of each phase to the full to explore various optical and electronic physical properties, including not only optical properties but also semiconductor properties," says Assistant Professor Arakawa.
This work was partly supported by JSPS KAKENHI Grant numbers 15H06285 and 17K14493, Toyota Physical and Chemical Research Institute Scholars and research grants from the Nitto Foundation, the Toukai Foundation for Technology and Research Foundation for the Electrotechnology of Chubu.
Patent
Yuki Arakawa, Satoyoshi Inui and Hideto Tsuji. Japanese Patent Application No. (2018) 84511
Technical Terms
- 1) A substitutional group in which sulfur (S) and an alkyl chain (CmH2m+1) are bonded
- 2) A system with delocalized π electrons, created by a series of alternate single and unsaturated bonds
- 3) A molecular structure in which two benzene rings are connected with a triple bond
- 4) An analogue in which sulfur is replaced with oxygen
Reference
Yuki Arakawa, Satoyoshi Inui and Hideto Tsuji. "Novel diphenylacetylene-based room-temperature liquid crystalline molecules with alkylthio groups, and investigation of the role for terminal alkyl chains in mesogenic incidence and tendency." Liquid Crystals, 2017.
http://dx.doi.org/10.1080/02678292.2017.1383521
硫黄を導入したπ共役系棒状液晶分子の開発
By 荒川 優樹豊橋技術科学大学の荒川優樹助教らは、硫黄成分を有するアルキルチオ基注1)を導入したπ-共役系注2)棒状分子の液晶化に成功し、室温を含む温度範囲で流動性の高いネマチック液晶を示す高複屈折性分子を開発しました。この分子設計により、液晶ディスプレイの高画質化などに寄与する新しい液晶材料としての応用が期待されます。
複屈折や誘電率(異方性)の大きな液晶材料は、主に液晶ディスプレイの低駆動電圧化、応答速度の向上などに寄与しております。さらには近年、様々なアプローチにより高複屈折性液晶材料は、輝度向上フィルムなどに用いられる広帯域円偏光反射フィルムや、連続発振を目指すコレステリック液晶レーザーに使われるなど、その応用例は増えております。
液晶材料を開発する上で実用性を考えると、室温で液晶相を形成するか、液晶の配向状態を固定する必要があります。しかしながら、複屈折や誘電率の向上には異方的な分子構造を有し、かつ電子リッチであることが必要条件であるため、大きな分子間力による相転移温度(特に融点)の向上は避けられません。つまり、室温では液晶状態を形成しにくくなります。
荒川優樹助教らは、温泉などの成分にも含まれ、日本の数少ない余剰資源である「硫黄」を含むアルキルチオ基(SCmH2m+1)を導入した分子系における液晶材料の研究を行ってきました。アルキルチオ基は分極率が高く、複屈折の向上に有効な置換基であることが期待されますが、一方で、アルキルチオ基を導入した棒状分子は液晶性を形成しにくく、π—共役系棒状分子において液晶性を示した報告例はほとんどありません。
これまでに、図1に示す、アルキルチオ基を導入したジフェニル-アセチレン構造(三重結合数が1)注3)の、片末端に炭素数5以上の十分な長さのアルキル鎖を導入することで、冷却過程において液晶性が発現され、さらには一部の誘導体は室温以下までその液晶相が過冷却されることを明らかにしました。これは、反平行に配列した分子間において、ベンゼン環の面外方向に安定なコンフォメーションを有するアルキル基(CnH2n+1)によりその分子間のパッキングが緩和され、配向を維持したまま分子の回転および併進運動(分子の位置の融解)が可能となることで液晶相が形成され、アルキルチオ基の大きな屈曲や低い電子供与性により転移温度が低温化するものと考えられます。また、長いアルキル基を導入した上で、アルキルチオ基の炭素数を変えることにより、粘性の高いレイヤー構造を有する高次のスメクチック相から、特に光学用途に重要な粘性の低いネマチック(N)相の形成までを作り分けることが可能となります。これらは、酸素類縁体注4)との比較でも光学特性が大幅に向上していることも確認されました。また最近では、上記分子設計指針を基に、三重結合を一つ拡張したジフェニル-ジアセチレン構造(n=2)における室温液晶、ならびに複屈折が最高で0.3に到達する高複屈折性液晶材料の開発にも成功しております。
「棒状構造の分子にアルキルチオ基を導入した液晶分子の報告例は極めて少なく、なぜ液晶相を形成しにくいのか、その原因も含めて、それら分子の特徴を明らかにした研究例はありませんでした。今後はそれぞれの相の特徴を最大限に生かし、光学特性だけでなく、半導体特性など、様々な光・電子物性の開拓も目指して参ります。」と荒川助教は述べています。
本研究は、以下の助成を受けて行われました。
- JSPS 科研費 Nos. 15H06285, 17K14493
- 公益財団法人豊田理化学研究所「豊田理研スカラー」
- 公益財団法人日東学術振興財団研究助成金
- 一般財団法人東海産業技術振興財団研究助成金
用語解説
- 注1) 硫黄に(S)にアルキル鎖(CmH2m+1)が結合した置換基
- 注2) 単結合と不飽和結合が交互に連結し、π電子が非局在化した系
- 注3) 二つのベンゼン環が三重結合で連結した分子構造
- 注4) 硫黄が酸素に置き換わった類縁体
Researcher Profile

| Name | Yuki Arakawa |
|---|---|
| Affiliation | Department of Environmental and Life Sciences |
| Title | Assistant Professor |
| Fields of Research | Synthetic organic chemistry, Polymer chemistry, Liquid crystals, Optical materials |
ここでコンテンツ終わりです。
