
ここからコンテンツです。

Structure of Electrolyte Controls Battery Performance
Toward implementation of calcium-ion batteries By Yoshiaki Murata
A research team at the Department of Electrical and Electronic Information Engineering, Toyohashi University of Technology has revealed that adding water into electrolyte improves the function of vanadium oxide, which is one of positive electrode materials used in calcium-ion batteries. Although water in electrolytes is known to produce many negative effects, it has now been found to bring about a phenomenon that speeds up the conventionally slow reaction of calcium-ion batteries. The results of the present study indicate that this phenomenon is caused by changes in the electrolyte structure. It is believed that this discovery will greatly benefit the development of electrolytes for implementing calcium-ion batteries in the future.
Secondary batteries are valuable resources that support various industries. Nowadays, secondary batteries are required to be even more powerful to cope with increased use of reusable energy and electric vehicles. Lithium-ion secondary batteries are already widely used as powerful secondary batteries. However, in recent years, the safety of secondary batteries has been brought into question with countless reports citing combustion. Going forward, the need for batteries in our current society is expected to increase exponentially along with the rise in electric vehicles. This means a higher demand for lithium, which in turn is expected to cause problems such as higher prices and potential resource depletion.
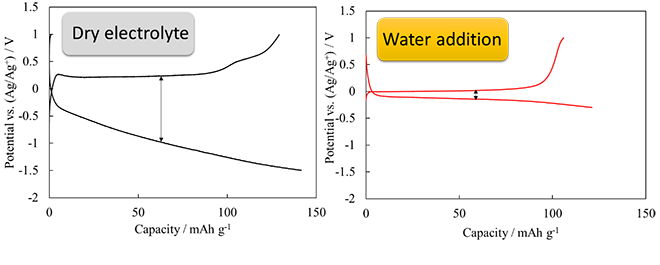
Calcium-ion batteries are a type of next-generation secondary battery that do not use lithium and can achieve a battery voltage that rivals that of lithium-ion batteries. Compared to lithium-ion batteries, calcium-ion batteries are safer, cheaper to produce and their resources are much more plentiful. While calcium-ion batteries are currently attracting attention for these reasons, they are still subject to a number of issues. One such issue is that they operate at a speed much lower than that of lithium-ion batteries. In this study, Toyohashi University reported that the slow operating speed of calcium-ion batteries could be improved by adding water into the electrolyte. The graphs of the test results show that overvoltage that occurs during charge/discharge greatly decreases as the amount of added water increases and that reaction proceeds without any problems. As a result of various tests, it was proved that this phenomenon is caused by the fact that the structure of the electrolyte is greatly changed by the addition of water.
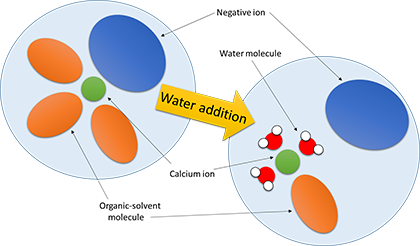
Yoshiaki Murata, PhD student and first author of the study explains that, "The electrolyte is made up of positive ions (calcium ions), negative ions and solvent molecules, and the state around the calcium ion greatly changes when water is added. What that means is that, in order to improve the performance of a calcium-ion battery, preferably no negative ion is attached to the calcium ion in the electrolyte and a solvent molecule that easily separates is attached to the calcium ion. While we still need to discover an electrolyte with these characteristics that does not include water in order to achieve calcium-ion batteries, the discovery of this phenomenon will surely help with future electrolyte development.
The result of the present study was actually a secondary result obtained while studying new electrolytes. Electrolytes need to be sufficiently dehydrated when they are developed, but this dehydration process is difficult. The present study arose when it was noticed that the characteristics of a battery which had an insufficiently-dehydrated electrolyte actually improved during tests. Although there have been reports of a phenomenon in which the performance of magnesium-ion batteries for example had improved after the addition of water, the mechanism behind this was not clearly understood. It was surprising that the same phenomenon could be seen in calcium-ion batteries, and we believe that elucidating the mechanism behind this behavior will prove useful for the future development of electrolytes.
Our research team is looking to develop and assess new electrolytes based on this newly discovered electrolyte structure that improves the performance of calcium-ion batteries. Further, we have not been the only ones to study this; there has been rapid increase in the number of studies of calcium-ion batteries in recent years. Ultimately, we would like to develop a calcium-ion battery that has the capability to rival or overtake lithium-ion batteries.
This work was partly supported by JSPS KAKENHI Grant Numbers JP24360109, JP18H01427 from the Japan Society for the Promotion of Science (JSPS).
Reference
Yoshiaki Murata, Shoki Takada, Tomohiro Obata, Tomohiro Tojo, Ryoji Inada, Yoji Sakurai (2019). Effect of water in electrolyte on the Ca2+ insertion/extraction properties of V2O5, Electrochimica Acta.
http://dx.doi.org/10.1016/j.electacta.2018.10.103
電解液の構造が電池の性能を支配する
カルシウムイオン電池の実現に向けてBy 村田 芳明
豊橋技術科学大学電気・電子情報工学系の研究チームは、カルシウムイオン電池の正極材料の一つである酸化バナジウムの性能が、電解液に水を添加することで向上することを明らかにしました。電解液に含まれる水は、様々な悪影響を及ぼすことが知られていますが、従来非常に低速だったカルシウムイオン電池の反応が高速化される現象が見出されました。今回の研究結果から、この現象は、電解液の構造の変化によって引き起こされていることが明らかとなりました。今後、カルシウムイオン電池実現のための電解液開発に向けて、重要な指標を与えると考えられます。
二次電池は、再生可能エネルギーの利用普及や、電気自動車の普及を支える重要な技術として、更なる性能の向上が必要です。性能の良い二次電池として、リチウムイオン電池が既に広く普及しています。その一方で、近年発火事故の報告は絶えず、二次電池の安全性は社会問題化しています。また、これからの社会では電池の需要は電気自動車の普及と共に爆発的に増加すると予想され、これによって生じるリチウムの需要増加は、資源枯渇リスクや価格高騰の観点で考慮すべき問題です。
カルシウムイオン電池は、リチウムを用いない次世代二次電池の一つです。カルシウムは次世代二次電池の中でも特に、リチウムイオン電池に匹敵する電池電圧を実現可能であり、リチウムイオン電池と比較して安全性が高く、資源量も豊富で、低コストであることから注目を集めています。しかしながら、カルシウムイオン電池は未だ多くの問題を抱えています。その一つが、リチウムイオンと比較してカルシウムイオンの動きが非常に低速なことです。
研究では、電解液に水を添加することで、この遅い動きを改善することを報告しました。実験結果の図から、放電・充電時に発生する過電圧が、水添加によって大きく減少し、反応がスムーズに進行していることが分かります。様々な検討の結果、この現象が電解液の構造が、水の添加によって大きく変化していることが原因となって引き起こされていることが明らかとなりました。
「電解液は、プラスイオン(カルシウムイオン)、マイナスイオン、溶媒分子で構成されており、水を添加した際には、カルシウムイオンの周りの状態が大きく変化します。具体的には、カルシウムイオン電池の性能を向上させるには、電解液の中でカルシウムイオンにマイナスイオンが付いておらず、さらには剥がれやすい溶媒分子が付いていることが好ましいです。カルシウムイオン電池を実現するためには、水を含まずにこれらの特徴を有する電解液を発見することが必要ですが、これらの現象を発見できたことはこれからの電解液開発にも必ず役立つはずです。」と筆頭著者である博士後期課程の村田芳明は説明します。
今回の成果は、新しい電解液の検討中に副次的に得られた成果です。電解液の開発の際には、電解液を十分に脱水する必要があります。脱水方法が難しく、脱水が不十分な状態の電解液を試験した際に、電池の特性が向上したのがこの研究の始まりです。水を添加した際に電池の性能が向上するという現象は、マグネシウムイオン電池などでは報告されていましたが、未だその機構が正確には解明されていません。カルシウムイオン電池でも同様の現象が起こった、という驚きと共に、この現象が発生する機構を解明すれば、これからの電解液開発に役立つと考えました。
研究チームは、今回発見できたカルシウムイオン電池の性能を向上させる電解液構造をもとに新たな電解液を開発・評価したいと考えています。また、我々の研究だけではなく、ここ数年でカルシウムイオン電池の研究例は急速に増加しつつあります。最終的には、リチウムイオン電池に匹敵、あるいはリチウムイオン電池を上回る性能を持ったカルシウムイオン電池の開発を行っていきたいと考えています。
Researcher Profile

| Name | Yoshiaki Murata |
|---|---|
| Affiliation | Department of Electrical and Electronic Information Engineering |
| Title | PhD student |
| Fields of Research | Energy Conversion Engineering / Electrochemistry |
ここでコンテンツ終わりです。
